Hydrogen HSE
This blog is based on the post Hydrogen HSE: Health, Safety and Environmental.
This post summarizes some of the HSE (Health, Safety and Environmental) issues that need to be considered when manufacturing, transporting, storing and using hydrogen.
Safety Diamond
The safety diamond for hydrogen is shown below.
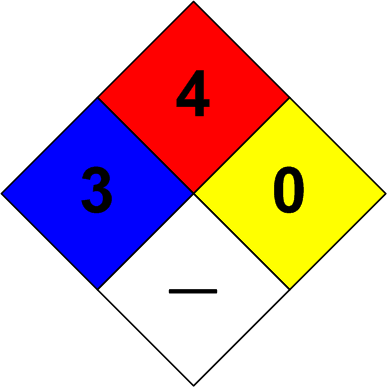
The numbers and colors have the following meaning.
-
Flammability (Red) — 4. Extremely flammable gas.
-
Instability (Yellow) — 0. Normally stable. Does not react with water.
-
Specific Hazards (White). None.
-
Health (Blue) — 3. Serious.
The legend associated with this Safety Diamond reads,
Colorless, odorless, highly flammable gas. Stored as compressed gas in cylinders. Simply asphyxiant (reduced oxygen available for breathing). Eye and skin contact with compressed gas may cause frostbite.
Health
With regard to health there are no special issues other than its asphyxiation potential if it displaces breathing air, and the potential for frostbite.
Safety
The following safety issues to do with hydrogen should be considered.
-
Hydrogen has a wide flammable range in air, which means that it can ignite more easily than other fuels. It can self-ignite when it leaks from a container at high pressure. Hydrogen also has a low ignition energy. (the reverse Joule-Thomson effect applies to hydrogen at normal operating temperatures) Therefore, ventilation and leak detection are particularly important.
-
A hydrogen flame is nearly invisible, particularly in bright daylight. Special flame detectors may be required.
-
Hydrogen has a zero rating for reactivity or toxicity. The letters ‘SA’ can be used here, meaning that the gas can exclude oxygen from the air at a particular location, and so create a ‘simply asphyxiation’ hazard.
-
Some metals can become brittle when exposed to hydrogen.
-
Liquid hydrogen has safety issues common to all cryogenic liquids.
-
High temperature hydrogen attack can cause metallic failure.
An overview of hydrogen safety and leak detection is provided by the Chemical Engineering magazine. (Jenkins, 2020).
Environmental
Hydrogen does not have any significant environmental concerns. If released it immediately escapes to the upper atmosphere. (It does, however, have a significant greenhouse gas impact.)
